Selected Research Highlights from Laboratories Affiliated with EMSSI
 Title of Project: Feeling States and Heart Rates; A Translational Study
Title of Project: Feeling States and Heart Rates; A Translational Study
Principle Investigator : Margaret Schneider, Ph.D.
Email: Interested in more information
Overview: By virtually all metrics (self-report, ambulatory monitoring, observation), most adolescents in the United States do not achieve levels of physical activity sufficient for optimal health and well-being. The debilitating adverse health effects of physical inactivity such as obesity, type 2 diabetes, and increased cardiovascular disease risk are, unfortunately, far more common among lower SES children and adolescents. It is also worrisome that rates of participation have not increased any meaningful amount despite growing recognition of the problem and attempts to promote greater levels of activity. New approaches are needed. This research builds on basic research into adolescent psychophysiology to test a novel intervention to promote increased physical activity among less-active adolescents.
This project extends discoveries we have made during the past two decades of NIH funding for our research investigating the mechanisms and health effects of physical activity behavior change among adolescents. Based on this earlier work, we have developed methods for identifying adolescents who are at higher risk for becoming sedentary as they mature. Our current study will be testing whether tailoring an intervention for these at-risk adolescents will be an effective means of keeping them from reducing their activity levels as they mature.
To carry out the specific aims of this project, we are working with Marshall Academy of the Arts in Long Beach to recruit a total of 140 middle-school students, each of whom will be in the study for two years. We have transformed a classroom at the school into a clinical laboratory where we conduct cardiorespiratory fitness assessments, electroencephalograms, and additional exercise assessments particular to this study. Assessments are conducted in the fall and spring, and the intervention is implemented during the winter. The project is a partnership between the research team and the physical education program at the school, and embodies many of the principals of Community-Based Participatory Research. Each semester, from 5-10 UCI undergraduate students gain research experience as Research Assistants on the study, many of whom present their projects at the Undergraduate Research Opportunity Program (UROP).
Information on Enrolling in this Study can be found HERE
==============================================================================
 Title of Project: Functional and Locomotor Consequences of ECM Remodeling
Title of Project: Functional and Locomotor Consequences of ECM Remodeling
Principle Investigator : Manny Azizi PhD
Email: Interested in more information from Dr. Azizi’s lab
Overview: An increase in the density and stiffness of extracellular matrix (ECM) of muscle is associated with aging as well as a broad range of neuromuscular disorders, including cerebral palsy, muscular dystrophy, and muscular responses secondary to stroke or spinal cord injury. These changes can lead to muscles becoming fibrotic and causing an increase in passive muscle stiffness. The primary aim of our work is to establish a mechanistic link between the remodeling of the muscle ECM and functional deficiencies of the locomotor system. This work aims to understanding how muscle fibrosis affects the length changes and mechanical work of muscles. We also test the hypothesis that muscle fibrosis will increase the likelihood of muscle damage and injury when muscles are actively stretched to dissipate mechanical energy. Finally, we use a modeling approach to relate changes in the passive stiffness of fibrotic muscle to a reduction in range of motion and gait deficiencies.
 In this work we use aging in rats as our model of ECM remodeling. Specifically, we will use a cross between F344 and the Norway brown rat (F344xFBN). This strain was specifically developed by the National Institute of Aging and is widely considered the premiere animal model system for aging studies. We integrate studies at the level of tissues, organs and organism to understand the functional and locomotor consequences of muscle fibrosis. Although this research is focused on the basic science of ECM remodeling, our results will likely have important implications for therapeutic and rehabilitation approaches for improving mobility in populations suffering from muscle fibrosis. This work is done in collaboration with Brown University and Cal State San Bernardino.
In this work we use aging in rats as our model of ECM remodeling. Specifically, we will use a cross between F344 and the Norway brown rat (F344xFBN). This strain was specifically developed by the National Institute of Aging and is widely considered the premiere animal model system for aging studies. We integrate studies at the level of tissues, organs and organism to understand the functional and locomotor consequences of muscle fibrosis. Although this research is focused on the basic science of ECM remodeling, our results will likely have important implications for therapeutic and rehabilitation approaches for improving mobility in populations suffering from muscle fibrosis. This work is done in collaboration with Brown University and Cal State San Bernardino.
==============================================================================
 Title of Project: Building Muscle by Restricting Blood
Title of Project: Building Muscle by Restricting Blood  Flow?
Flow?
Principle Investigator : Bruce Tromberg, PhD
Email: Interested in more information from Dr. Tromberg’s group
Overview: It has been shown by other researchers that muscle strength and size can be enhanced by performing exercise with some degree of pressure applied to the limb to restrict blood flow. The mechanism of blood flow restriction (BFR) is still being studied, and the techniques is being applied to many different populations who have difficulty tolerating intense, heavy-load exercise. Still, there is not much known about how BFR affects muscle oxygen use and blood flow dynamically.
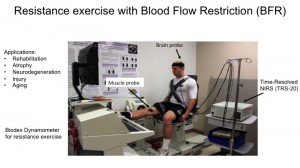 We used time resolved near infrared spectroscopy (TR-NIRS) to measure the effect of BFR on exercise in both the muscle and the brain. TR-NIRS uses light on the surface of the skin to quantitatively measure levels of blood, and its oxygen content, expressed as concentrations of oxyhemoglobin [HbO2], deoxyhemoglobin [HbR], total hemoglobin [THb], and Oxygen saturation (stO2). (click on image to enlarge)
We used time resolved near infrared spectroscopy (TR-NIRS) to measure the effect of BFR on exercise in both the muscle and the brain. TR-NIRS uses light on the surface of the skin to quantitatively measure levels of blood, and its oxygen content, expressed as concentrations of oxyhemoglobin [HbO2], deoxyhemoglobin [HbR], total hemoglobin [THb], and Oxygen saturation (stO2). (click on image to enlarge)
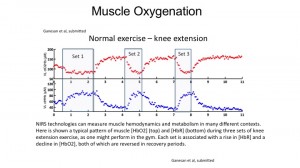 Using this technique, we found that BFR has significant effects on muscle oxygenation and blood flow, specifically during recovery from exercise. This change in oxygen extraction during recovery might lead to the benefits that have been observed using BFR. Additionally, we also found that there was elevated Oxygen extraction in the prefrontal cortex, a part of the brain involved in attention and exertion. This could be related to the fact that exercise with BFR is much more difficult to complete than the same exercise without BFR. These results demonstrate a an interesting application for non invasive NIRS technology in monitoring exercise therapy. (click on image to enlarge)
Using this technique, we found that BFR has significant effects on muscle oxygenation and blood flow, specifically during recovery from exercise. This change in oxygen extraction during recovery might lead to the benefits that have been observed using BFR. Additionally, we also found that there was elevated Oxygen extraction in the prefrontal cortex, a part of the brain involved in attention and exertion. This could be related to the fact that exercise with BFR is much more difficult to complete than the same exercise without BFR. These results demonstrate a an interesting application for non invasive NIRS technology in monitoring exercise therapy. (click on image to enlarge)
